What Is the Best Way to Estimate the Map?
Abstruse
To assess the differences amidst seven different methods for the calculation of mean arterial pressure level (MAP) and to identify the formula that provides MAP values that are more closely associated with target organ deterioration as expressed by the carotid cross-sectional area (CSA), carotid-to-femoral pulse-wave velocity (cf-PWV) and left ventricular mass (LVM). The study population consisted of 1878 subjects who underwent noninvasive cardiovascular take a chance cess. Blood pressure (BP) was assessed in all subjects, and MAP was calculated past direct oscillometry and 6 different formulas. Carotid artery ultrasound imaging was performed in 1628 subjects. The CSA of the right and left common carotid artery (CCA) were calculated and used as surrogates of arterial wall mass and hypertrophy. Aortic stiffness was evaluated in 1763 subjects by measuring the cf-PWV. Finally, 218 subjects underwent echocardiographic examination for the assessment of LVM. Amongst the examined methods of MAP calculation, the formula MAPone=[diastolic BP]+0.412 × [pulse pressure] yielded the strongest correlations with the LVM, cf-PWV and CSA of the right and left CCA, even after adjusting for age and gender. The MAP calculation using the 0.412 was superior compared with the traditional formula that uses the 0.33 for the discrimination of subjects with left ventricular and carotid wall hypertrophy, every bit well as subjects with increased aortic stiffness. MAP estimated with the 0.412 is better correlated with target organ deterioration compared with other formulas. Future studies are needed to explore the accuracy of these formulas for MAP estimation compared with directly intra-arterial BP measurement.
Introduction
Mean arterial claret pressure level (MAP) is the major hemodynamic determinant of tissue perfusion regardless of pulse pressure, and information technology is also a key parameter that influences cardiac role and the wall backdrop of fundamental arteries. Higher MAP levels are related to cardiovascular (CV) disease and target organ damage, whereas low levels may exist detrimental in hemodynamically unstable, critically ill patients. Beyond the pathophysiological and clinical relevance of MAP, there are many circumstances in which a MAP calculation is required, such as the determination of peripheral vascular resistance, partial pulse pressure, calibration of devices that judge central blood pressure (BP) and others.one, ii, 3, four
Undoubtedly, it is essential to estimate MAP values accurately. The gold-standard method for the measurement of the 'actual' value of MAP is the adding of the surface area nether the BP waveform, as determined by the time-averaged BP values over the cardiac wheel, which is recorded invasively past catheter-manometer systems. However, in routine clinical practice, MAP is estimated non-invasively either by using the internal proprietary algorithms of automated oscillometric sphygmomanometers or past mathematical formulas. The accuracy of MAP estimation using oscillometric devices is rarely reported in the literature, and most importantly, several commercial devices do not study MAP, simply use information technology to calculate the systolic and diastolic BP via proprietary algorithms.
Pressure waveforms differ along the arterial tree due to wave propagation and reflection phenomena. The change in the shape of pressure waves can be quantified via the and then-called 'course cistron,' which characterizes the ratio of the difference betwixt its hateful and minimum values over the amplitude of a wave.5 Therefore, the form gene non only quantifies the shape of the wave, but it besides expresses the percentage of pulse pressure level to add together to diastolic blood pressure (DBP) to estimate MAP.
On the basis of the concept of 'form gene,' several equations take been proposed for the adding of MAP, as reported in Tabular array 1. In 1939, Wezler and Bögerr6 proposed the formula MAP=0.42 × SBP+0.58 × DBP, where SBP is the systolic and DBP is the diastolic BP. Meaney et al. seven proposed an culling expression of the above formula equally follows: MAPi=DBP+0.412 × PP, where PP is the pulse pressure. The most common and widely used formula for MAP calculation is that proposed by Gauer in 1960:8 MAP2=DBP+0.33 × PP. In 1999, Chemla et al. 9 proposed an improvement for the traditional formula, with MAPthree=DBP+0.33 × PP+5 mm Hg. Razminia et al. 10 in 2004 included the center charge per unit in the equation for MAP calculation equally follows: MAP4=DBP+[0.33+(0.0012 × HR)] × PP. In essence, these formulas differ in the coefficient used to integrate PP in the algorithm, which actually represents the form factor of the force per unit area waveform.two Chemla et al.,xi in 2005, proposed a different mathematical equation based on the product of SBP by DBP: MAP5=(SBP × DBP)½. MAP can also be calculated past the integration of pressure waveforms recorded non-invasively (that is, applanation tonometry) and calibrated by brachial SBP and DBP values (MAP6).12 Finally, in the present analysis, the 'original' MAP (MAP7), equally provided by the proprietary algorithm of a commercially bachelor automated oscillometric device, was also examined.
Currently, there is no consensus regarding which formula yields the most accurate MAP estimation, although MAPtwo is the most popular and widely used equation. In addition, there is a lack of prove regarding which of these different MAP estimates is ameliorate associated with target organ harm. The objective of this report was to assess the differences among the original MAP values provided by a commercially available automated oscillometric device and the diverse MAP levels calculated using different formulas (Table 1). We also determined which method provides MAP values that are more closely associated with target organ characteristics and deterioration as expressed past the left ventricular mass, carotid wall thickness and stiffness of the aorta. Because aging and gender significantly attune the course factor of the pressure waveform, these two parameters were taken into detail consideration in the present assay.
Methods
Study population
The report population consisted of 1878 subjects (Caucasians) who were referred to the Cardiovascular Research Laboratory of the Outset Deptartment of Propaedeutic and Internal Medicine, Laiko University Hospital for CV risk assessment due to the presence of either classical and/or novel chronic inflammatory diseases. The population characteristics are shown in Table 2. All subjects had a normal sinus rhythm. Subjects with arrhythmia and astringent obesity (body mass index >twoscore kg m−2) were excluded from the written report because applanation arterial tonometry and pulse-wave analysis were not feasible or had an unacceptable quality. All subjects were examined in a placidity, temperature-controlled environment (22–25 °C). Co-ordinate to laboratory routine practise, patients nether pharmaceutical treatment were brash to abjure from their medication for at least 10 h prior to the examination, as well as to abstain from booze, caffeinated beverages or any other vasoactive substances for at least iii h. The written report protocol complied with the declarations of Helsinki and was approved past the institutional scientific committee. All subjects gave informed consent before inbound the study.
Blood force per unit area measurement at the brachial artery
Brachial BP was assessed at the supine position after at least 10 min of residue. Triple brachial BP recording was performed (with ane-min interval between readings) in the correct arm with a validated automated oscillometric device (Microlife WatchBP Office, Microlife AG, Widnau, Switzerland).thirteen The boilerplate value of the three BP readings (for SBP and DBP) was used for the calculation of MAP values using the examined methods (Table i).
Not-invasive recording of continuous blood pressure waves
MAP was too determined by the integration of calibrated radial force per unit area waves recorded past applanation tonometry, which was performed immediately subsequently the BP triple measurement. In particular, continuous pressure level waves were recorded by a high-allegiance tonometer (Millar Instruments, Houston, TX, The states), and an averaged single pressure wave was adamant using the Sphygmocor Software (Atcor Medical, West Ryde, Australia). In add-on, the average value for the center charge per unit was determined by the system based on the cardiac periods of the recorded force per unit area waves, and this value was used for the adding of MAP4. The acquired tonometric waves were calibrated using the respective SBP and DBP levels measured at the brachial avenue. The calibration of radial pressure waves using brachial SBP and DBP measured past cuff oscillometry assumes that the brachial BP is equal to the radial BP. Nonetheless, this supposition may non be authentic considering it is possible to take an amplification of the SBP betwixt the brachial and radial artery.five
Left ventricular mass assessment
Left ventricular mass (LVM) and hypertrophy (LVH) were assessed every bit previously described.14 In brief, transthoracic echocardiography was performed in all patients by the same operator using a high-end ultrasound system (Vivid 7 Pro, Full general Electric, Fairfield, CT, Us) in accordance with the American Social club of Echocardiography (ASE) and European Association of Echocardiography guidelines and recommendations.15 Measurements for the Yard-mode-guided calculation of LVM were recorded in the parasternal short-axis view. LVM was further standardized to body size, providing the LVM alphabetize (LVMI). LVMI was calculated by the ratio of LVM to body surface area (BSA), where BSA=[(weight × height)/3600]0.five.
Carotid wall hypertrophy
Carotid wall CSA, instead of intima-medial thickness, was used as a more comprehensive index of the arterial wall volume or mass because it is closer to arterial hypertrophy16 using a previously applied formula.17 Briefly, the common carotid artery was scanned for the presence of plaques. The average intimal-medial thickness of a plaque-complimentary segment of the mutual carotid artery (ane cm proximally to the bifurcation) was measured past a defended inbuilt software (Vivid vii Pro). 2 sequential CSA of the same mutual carotid segment were analyzed, and their hateful value was used in the analysis.
Cess of aortic stiffness
Aortic stiffness was assessed by measuring the carotid-to-femoral pulse-wave velocity (cf-PWV) every bit previously described.xviii In brief, cf-PWV is calculated by the ratio of the estimated pulse transit fourth dimension and the distance travelled past the pressure wave between the two recording sites. Several methods be for pulse transit time estimation, which may often yield divergent results.19 In this study, we used the tangential (or intersecting tangents) method, which was implemented at the SphygmoCor organisation (AtCor Medical, Sydney, Australia). At showtime, the travel altitude of the pressure wave was determined as the distance from the suprasternic notch to the femoral artery minus the distance from the carotid artery to the suprasternic notch. And then, arterial pressure waves were recorded by applanation tonometry using a high-allegiance hand-held tonometer (SPT-301, Millar Instruments). Pressure waves were showtime recorded at the carotid avenue and then, within a few seconds, at the femoral artery. The time delay between the two waves (transit time) was determined using registration with a simultaneously recorded ECG. All recordings were made at the supine position. At least two repeated measurements of PWV were performed, and their average value was used in the assay every bit previously recommended.20
Assessment of target organ deterioration
The following indices were considered as indicators of target organ deterioration:
-
LVH defined as LVMI values >95 chiliad one thousand−ii in women or >115 thou k−2 in men using the ASE formula.fifteen
-
Increased aortic stiffness, defined as cf-PWV >10 m s−1.21
-
Carotid wall hypertrophy, defined arbitrarily by the quaternary upper quartile of CSA-RCCA or CSA-LCCA values.
All vascular studies were performed by the same experienced operator using the same device for each examination.
Statistical analysis
The understanding between different MAP estimations was assessed using the Bland and Altman analysis.22 Co-ordinate to this method, the differences between the two measurements (bias)=(MAPi−MAPj) are plotted confronting their mean value, (MAPi+MAPj)/ii. The s.d. of the differences between the two dissimilar MAP estimates was also determined. Furthermore, we used the Pearson correlation coefficient (r), intraclass correlation coefficient (ICC) and coefficient of variation (CVar) to assess the understanding, consistency and variation between unlike MAP estimates. These statistical methods and parameters were described thoroughly elsewhere.23, 24 The bivariate association of each MAP judge with parameters assessing the target organs (LV and carotid wall mass likewise equally cf-PWV) was evaluated using the Pearson correlation coefficient. Farther multivariate regression models were used to adjust the relationship of MAP with each parameter of target organ deterioration for age and gender. Williams' statistic, which is often noted every bit Steiger's Z-test, was used to compare the correlation coefficients of cardiovascular parameters with MAP values. Receiver operating feature (ROC) curve analysis was used to determine the comparative ability of different MAP estimates to discriminate subjects with target organ damage. Areas under the ROC curves were compared based on the methodology proposed by DeLong, DeLong and Clarke-Pearson.25 All tests were two-tailed, and statistical significance was indicated by P-values<0.05. Statistical assay was performed using SPSS xx (IBM Corp., Armonk, NY, USA).
Results
The demographic, clinical and hemodynamic characteristics of the study population are reported in Tabular array 2. The MAP values estimated using the vii different formulas/methods were compared with each other. The statistical parameters indicating variation and understanding between unlike MAP estimates are shown in Table 3. The mean±s.d. value of the MAP values adamant past each formula is depicted in Figure 1. The mean differences and s.d. of differences between each pair of dissimilar estimates for MAP are presented in Figure 2.
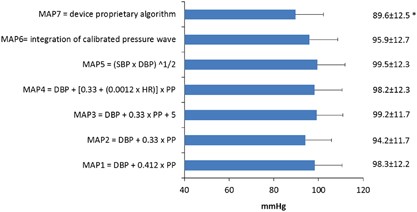
Mean value±s.d. of mean arterial pressure level (MAP) estimated past each different method. *MAP values past using the Microlife WatchBP device was recorded in 872 subjects. A full colour version of this figure is bachelor at Hypertension Inquiry periodical online.
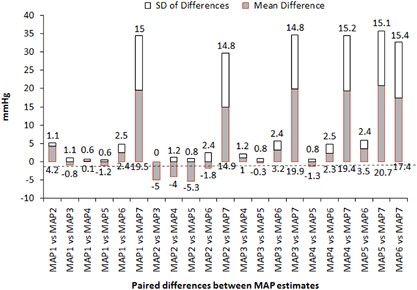
Hateful differences and s.d. of differences from paired comparisons between MAP estimates derived by unlike methods/formulas as defined in Table 1. A full color version of this figure is available at Hypertension Enquiry journal online.
Clan of each MAP estimate with cardiovascular parameters
The bivariate correlations of each MAP guess with LVMI, carotid wall mass and aortic stiffness are reported in Tabular array iv. All MAP estimates are correlated with all of the examined cardiac and vascular parameters (P<0.01). Among the seven different MAP estimates, MAPane yielded the strongest correlation with all three cardiovascular parameters. MAPseven provided the everyman correlation coefficient. Nosotros compared the correlation coefficient of LVMI with MAP1 (r=0.212) with the corresponding correlation coefficient of LVMI with the traditional MAP2 guess (r=0.202). The 2 correlation coefficients were marginally not significantly dissimilar (P=0.052). The correlation coefficients of MAPi with CSA-LCCA (r=0.35) and CSA-RCCA (r=0.365) were higher (P<0.001) than the respective coefficients of MAP2 (r=0.332 and r=0.344). Finally, the correlation of MAP1 with cf-PWV (r=0.441) was significantly stronger than the respective correlation (r=0.415) with MAPtwo (P<0.001). MAP1 yielded significantly higher correlation coefficients with LVMI, the CSA of the correct and left mutual carotid artery, as well as with cf-PWV compared with the MAP7 value estimated by the internal algorithm of the automated oscillometric device used in this study. The correlation coefficients of MAP1 with cardiovascular parameters were compared simply with those of MAP2 because the latter provided the closest results to the old compared with the remaining MAP3,4,5,6,vii values.
The relationship of each MAP estimate with the parameters of target organ damage was further assessed after adjusting for age and gender (Tabular array 5). MAP1 was, still, better associated with LVMI, CSA-LCCA, CSA-RCCA and cf-PWV compared with the traditional formula (MAP2).
Association of MAP1 and MAPtwo estimates with target organ deterioration
Assessment of the comparative ability of MAP1 and MAP2 to discriminate target organ deterioration was performed past ROC curve analysis. MAP1 had a greater ability than MAPii to discriminate LV hypertrophy and carotid wall hypertrophy (cross-sectional area at the fourth quartile of the studied population) as well equally aortic stiffness (defined by cf-PWV values greater than x thousand s−ane), every bit shown by the larger areas under the ROC curves (Tabular array 6).
Discussion
The nowadays study evaluated, for the outset time, the association of 7 different MAP estimates using various methods (formulas) with LVM, carotid wall mass and aortic stiffness. The formula proposed by Meaney et al., MAPane=DBP+0.412 × PP,vii provided MAP values ameliorate related to these cardiovascular parameters than the respective MAP values estimated using the classic formula, MAP2=DBP+0.33 × PP. Furthermore, MAP1 was a amend discriminator than MAP2 for subjects with left ventricular and carotid wall hypertrophy, also equally with increased aortic stiffness. The other MAP estimates (MAPiii,4,v,6,7) were less able to discriminate subjects with target organ deterioration.
Physiological and clinical relevance of mean arterial claret pressure
The mean arterial BP is the actual driving pressure for peripheral claret period and is, physiologically, a better indicator of perfusion to vital organs than systolic claret pressure. MAP values together with measures of cardiac output permit the estimation of peripheral resistance. When MAP is perturbed from the regulated level, cardiac output and total peripheral resistance are adapted to restore MAP back to the appropriate level. This regulation occurs due to the reflex of baroreceptors, which are located in the carotid sinus and also in the aortic arch and ventricles.
In some populations, MAP is a stronger determinant of LV structural features compared with other BP parameters.26 Moreover, in specific populations, MAP may exist more accurate in predicting cardiovascular prognosis than other BP parameters, such as systolic and diastolic force per unit area.27, 28 In addition, mean rather than systolic BP is the preferred metric in the intensive care unit to guide therapy.29 The superiority of MAP over the SBP was also observed in the BOSHI report,30 which compared measurements of maternal home BP with clinic BP earlier 20 weeks of gestation to determine associations with the risk of delivering a lower-nativity-weight infant. It was reported that high maternal home DBP and MAP, but non SBP, before 20 weeks of gestation was independently associated with a higher run a risk of lower babe birth weight than dispensary DBP and MAP.xxx
In clinical practice, in that location are several situations in which it is essential to monitor MAP levels. For instance, in patients with sepsis, vasopressors are oft titrated based on the MAP. According to the guidelines of the Surviving Sepsis Entrada,31 it is recommended that MAP should exist maintained ⩾65 mm Hg. In addition, treatment of patients with stroke or head injury may depend on the patient's MAP, whereas the admission MAP, as assessed using the class gene 0.3, in patients with stroke is an important parameter.32 Some other recent merely disquisitional application of MAP is the scale of devices for the noninvasive estimation of primal aortic BP.33, 34, 35 A common method for the calibration of recorded arterial pulse waves relies on the use of MAP and DBP values, which strongly bear on the accuracy of measurements.36
However, until at present, clinical practice has traditionally relied on the noninvasive auscultatory method to measure SBP and DBP. Past contrast, MAP constitutes the sole parameter physically measured by the most electric current oscillometric techniques, merely these values are often non reported. Thus, alternatively, MAP is calculated using a formula, most often i of those examined in this study. It should be noted, however, that current practice guidelines have been slow to integrate MAP evaluation in vital sign monitoring. For example, Cardiology and Hypertension Societies define hypertension based on SBP and DBP measurements only.37 By contrast, the Guild for Critical Intendance Medicine38 has utilized both systolic and mean arterial claret pressure for defining sepsis-induced hypotension, whereas MAP is used as a therapeutic target.
Association of MAP with target organ deterioration
In this study, we found that MAP is positively associated with LVM, which is consistent with previous findings.39, xl, 41 Nevertheless, the present written report showed that the MAP adding using a class factor of 0.412 is better related to LVM than MAP values derived from the form factor 0.33 (1/3). Furthermore, MAPone (vs. the traditional MAPtwo) was a better discriminator of subjects with LV hypertrophy. Similarly, all MAP values were positively related with the right and left carotid wall mass, which is consistent with previous studies,42, 43, 44 but again, MAP1 was the strongest determinant of the carotid wall mass. Finally, aortic stiffness, every bit assessed by carotid-to-femoral PWV, was positively related with MAP levels.45, 46 MAP1 yielded the highest correlation coefficient and was also a better discriminator of subjects with increased aortic stiffness (PWV>10 one thousand s−i). Of the vii examined methods for MAP interpretation, the internal proprietary algorithm of the utilized automated oscillometric device provided MAP values, which had the lowest association with the parameters of target organ impairment.
Notably, all formulas using a single form factor for the interpretation of MAP based on brachial BP measurements accept inherent flaws when applied to different populations. Nonetheless, in the Asklipeios Study,5 Segers et al. establish that the values of the form factor at the brachial avenue for different age groups and gender are close to the value of 0.4. Furthermore, there is a non-negligible amplification of SBP between the brachial and the radial artery,5 which suggests that the apply of the MAP6 method may include an additional source of error in MAP estimation. More chiefly, in a previous invasive written report providing intra-arterial pressures at the brachial avenue, information technology was observed that the mean pressure at the upper arm is underestimated when calculated using the traditional formula of calculation ane-3rd of the pulse pressure to the diastolic pressure level.47 Conversely, this underestimation can exist avoided by adding 40% of the pulse pressure to the diastolic pressure,47 suggesting that the utilize of the form cistron 0.4 yields more than authentic MAP estimations compared to invasive measurements. The above studies5, 47 further support our findings. Undoubtedly, invasive studies are needed to examine the accuracy of the various methods and techniques for the noninvasive estimation of MAP and larger scale studies are needed in different populations to further determine whether the most accurate MAP interpretation is translated to superior clinical relevance.
Limitations
This study did not evaluate the accurateness of each different MAP estimation, which could ideally exist achieved by comparison of the estimated MAP values with direct measured MAP through the intra-arterial, continuous recording of blood pressure waves. Therefore, substantial and thorough reasoning for which method of MAP estimation should be considered the almost accurate cannot be derived by the present analysis. Consequently, these findings regarding the highest correlation to target organ deterioration should not exist confused with ameliorate accuracy.
Unfortunately, LVM was assessed only in a minor number of patients (N=218) compared to the total population (North=1878). In improver, there were no bachelor data with which to test the clan of MAPvii with LVM.
Conclusions
In the existing literature, information technology is evident that MAP is most frequently calculated by the MAP2 formula. This formula is basically derived using the widely applied rule of thumb that assumes a class factor of ane-third (33%). However, it has been previously establish that 'this value is too low and that the 1-3rd rule to estimate mean arterial pressure should be reconsidered.'5
In many published studies, which formula was used for the MAP adding is often unknown. In the present study, nosotros demonstrated that the estimation of MAP using the form gene value 0.412 provides values that are more strongly related with cardiovascular parameters compared with MAP adamant by the traditional formula, which uses the course factor value 0.33. More than importantly, we found that the parameters of target organ deterioration, such as left ventricular and carotid wall hypertrophy as well as increased aortic stiffness, are better related with MAP1 than MAPtwo values. This show supports that MAP1 may take a superior diagnostic or even prognostic ability compared with the traditional formula (MAPtwo), but this should exist investigated further. In addition, future invasive validation studies are required to determine the accuracy of the different MAP estimates using the various bachelor formulas compared with intra-arterially measured mean arterial pressures.
References
-
Nakayama Y, Ueda H, Tsumura One thousand, Yoshimaru One thousand, Hayashi T . Ascending fractional pulse pressure closely relating to large avenue office. J Hum Hypertens 2002; 16: 243–247.
-
Salvi P . Pulse Waves: How Vascular Hemodynamics Affects Blood Pressure. Springer-Verlag. 2012.
-
Zhu Twoscore, Wang J, Jiang RZ, Teng YC . Pulsatility index in combination with biomarkers or mean arterial pressure level for the prediction of pre-eclampsia: Systematic literature review and meta-assay. Ann Med 2015; 47: 414–422.
-
Hsu CH, Chang JB, Liu IC, Lau SC, Yu SM, Hsieh CH, Lin JD, Liang YJ, Pei D, Chen YL . Mean arterial pressure is better at predicting future metabolic syndrome in the normotensive elderly: a prospective cohort report in Taiwan. Prev Med 2015; 72: 76–82.
-
Segers P, Mahieu D, Kips J, Rietzschel East, De Buyzere 1000, De Bacquer D, Bekaert S, De Backer 1000, Gillebert T, Verdonck P, Van Bortel L, Asklepios I . Distension of the force per unit area pulse in the upper limb in healthy, middle-aged men and women. Hypertension 2009; 54: 414–420.
-
Wezler K, Böger A . Dice Dynamik des arteriellen Systems: Der arterielle Blutdruck und seine Komponenten. Ergebn Physiol 1939; 41: 292–606.
-
Meaney East, Alva F, Moguel R, Meaney A, Alva J, Webel R . Formula and nomogram for the sphygmomanometric adding of the hateful arterial force per unit area. Eye 2000; 84: 64.
-
Gauer O . Kreislauf des Blutes. Lehrbuch der Physiologie des Menschen. Landois Fifty RH-U, editor: Von Urban and Schwarzenberg, 1960.
-
Chemla D, Hebert JL, Zamani K, Coirault C, Lecarpentier Y . Estimation of mean aortic pressure. Lancet 1999; 354: 596.
-
Razminia K, Trivedi A, Molnar J, Elbzour M, Guerrero M, Salem Y, Ahmed A, Khosla S, Lubell DL . Validation of a new formula for mean arterial pressure calculation: the new formula is superior to the standard formula. Catheter Cardiovasc Interv 2004; 63: 419–425.
-
Chemla D, Nitenberg A . A call for improving mean aortic pressure interpretation. Am J Hypertens 2005; eighteen: 891.
-
O'Rourke MF, Pauca A, Jiang XJ . Pulse wave analysis. Br J Clin Pharmacol 2001; 51: 507–522.
-
Stergiou GS, Tzamouranis D, Protogerou A, Nasothimiou E, Kapralos C . Validation of the Microlife Watch BP Office professional device for role blood pressure measurement according to the International protocol. Blood Press Monit 2008; 13: 299–303.
-
Protogerou AD, Argyris AA, Papaioannou TG, Kollias GE, Konstantonis GD, Nasothimiou East, Achimastos A, Blacher J, Safar ME, Sfikakis PP . Left-ventricular hypertrophy is associated better with 24-h aortic pressure than 24-h brachial pressure in hypertensive patients: the SAFAR study. J Hypertens 2014; 32: 1805–1814.
-
Lang RM, Bierig K, Devereux RB, Flachskampf FA, Foster Due east, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise J, Solomon S, Spencer KT, John Sutton Chiliad St, Stewart West American Society of Echocardiography's N, Standards C, Task Strength on Sleeping accommodation Q, American College of Cardiology Echocardiography C, American Eye A, European Association of Echocardiography ESoC. Recommendations for chamber quantification. Eur J Echocardiogr 2006; 7: 79–108.
-
Roman MJ, Devereux RB, Schwartz JE, Lockshin Physician, Paget SA, Davis A, Crow MK, Sammaritano L, Levine DM, Shankar BA, Moeller East, Salmon JE . Arterial stiffness in chronic inflammatory diseases. Hypertension 2005; 46: 194–199.
-
Roman MJ, Pickering TG, Schwartz JE, Pini R, Devereux RB . Relation of arterial structure and function to left ventricular geometric patterns in hypertensive adults. J Am Coll Cardiol 1996; 28: 751–756.
-
Laurent Southward, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, Pannier B, Vlachopoulos C, Wilkinson I, Struijker-Boudier H European Network for Non-invasive Investigation of Large A. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 2006; 27: 2588–2605.
-
Papaioannou TG, Vardoulis O, Protogerou A, Konstantonis G, Sfikakis PP, Stefanadis C, Stergiopulos Due north . In vivo evaluation of a novel 'diastole-patching' algorithm for the estimation of pulse transit time: advancing the precision in pulse wave velocity measurement. Physiol Meas 2015; 36: 149–161.
-
Papaioannou TG, Protogerou AD, Nasothimiou EG, Tzamouranis D, Skliros N, Achimastos A, Papadogiannis D, Stefanadis CI . Cess of differences betwixt repeated pulse wave velocity measurements in terms of 'bias' in the extrapolated cardiovascular chance and the classification of aortic stiffness: is a single PWV measurement enough? J Hum Hypertens 2012; 26: 594–602.
-
Mancia 1000, Fagard R, Narkiewicz M, Redon J, Zanchetti A, Bohm M, Christiaens T, Cifkova R, De Backer G, Dominiczak A, Galderisi M, Grobbee DE, Jaarsma T, Kirchhof P, Kjeldsen SE, Laurent S, Manolis AJ, Nilsson PM, Ruilope LM, Schmieder RE, Sirnes PA, Sleight P, Viigimaa M, Waeber B, Zannad F Job Force M 2013 ESH/ESC Guidelines for the management of arterial hypertension: the Job Force for the management of arterial hypertension of the European Lodge of Hypertension (ESH) and of the European Gild of Cardiology (ESC). J Hypertens 2013; 31: 1281–1357.
-
Banal JM, Altman DG . Statistical methods for assessing agreement betwixt two methods of clinical measurement. Lancet 1986; 1: 307–310.
-
Papaioannou TG, Argyris A, Protogerou AD, Vrachatis D, Nasothimiou EG, Sfikakis PP, Stergiou GS, Stefanadis CI . Non-invasive 24 hour ambulatory monitoring of aortic wave reflection and arterial stiffness by a novel oscillometric device: the first feasibility and reproducibility report. Int J Cardiol 2013; 169: 57–61.
-
Bartko JJ . The intraclass correlation coefficient as a measure of reliability. Psychol Rep 1966; 19: three–eleven.
-
DeLong ER, DeLong DM, Clarke-Pearson DL . Comparison the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 1988; 44: 837–845.
-
Rosendorff C, Become O, Schmeidler J, Silverman JM, Beeri MS . Correlation of arterial blood pressure and compliance with left ventricular structure and function in the very elderly. J Am Soc Hypertens 2012; vi: 48–55.
-
Avanzini F, Alli C, Boccanelli A, Chieffo C, Franzosi MG, Geraci East, Maggioni AP, Marfisi RM, Nicolosi GL, Schweiger C, Tavazzi L, Tognoni K, Valagussa F, Marchioli R . Investigators GI-P. Loftier pulse pressure level and low mean arterial pressure: 2 predictors of expiry after a myocardial infarction. J Hypertens 2006; 24: 2377–2385.
-
Zheng 50, Sun Z, Li J, Yu J, Wei Y, Zhang 10, Liu S, Li J, Xu C, Hu D, Dominicus Y . Mean arterial pressure: a improve mark of stroke in patients with uncontrolled hypertension in rural areas of China. Intern Med 2007; 46: 1495–1500.
-
Lehman LW, Saeed M, Talmor D, Marker R, Malhotra A . Methods of claret pressure measurement in the ICU. Crit Care Med 2013; 41: 34–40.
-
Iwama Northward, Metoki H, Ohkubo T, Ishikuro M, Obara T, Kikuya Thou, Yagihashi K, Nishigori H, Sugiyama T, Sugawara J, Yaegashi N, Hoshi K, Suzuki Grand, Kuriyama S, Imai Y Grouping BS. Maternal clinic and home claret pressure measurements during pregnancy and infant birth weight: the BOSHI study. Hypertens Res 2015; 39: 151–157.
-
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart One thousand, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb Due south, Beale RJ, Vincent JL, Moreno R . Surviving Sepsis Campaign Guidelines Committee including The Pediatric S. Surviving Sepsis Campaign: international guidelines for management of astringent sepsis and septic shock, 2012. Intensive Intendance Med 2013; 39: 165–228.
-
Eizenberg Y, Koton South, Tanne D, Grossman E . Association of age and admission mean arterial claret pressure level in patients with stroke-information from a national stroke registry. Hypertens Res 2016; 39: 356–361.
-
Papaioannou TG, Protogerou AD, Stamatelopoulos KS, Vavuranakis M, Stefanadis C . Non-invasive methods and techniques for key blood pressure estimation: procedures, validation, reproducibility and limitations. Curr Pharm Des 2009; 15: 245–253.
-
Salvi P, Grillo A, Parati G . Noninvasive estimation of central blood force per unit area and analysis of pulse waves by applanation tonometry. Hypertens Res 2015; 38: 646–648.
-
Martin JS, Borges AR, Christy JBt, Beck DT . Considerations for SphygmoCor radial artery pulse wave analysis: side selection and peripheral arterial blood pressure calibration. Hypertens Res 2015; 38: 675–683.
-
Laugesen Due east, Rossen NB, Peters CD, Maeng M, Ebbehoj E, Knudsen ST, Hansen KW, Botker HE, Poulsen PL . Assessment of central claret pressure in patients with type ii diabetes: a comparison between SphygmoCor and invasively measured values. Am J Hypertens 2014; 27: 169–176.
-
Mancia One thousand, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, Christiaens T, Cifkova R, De Backer G, Dominiczak A, Galderisi One thousand, Grobbee DE, Jaarsma T, Kirchhof P, Kjeldsen SE, Laurent S, Manolis AJ, Nilsson PM, Ruilope LM, Schmieder RE, Sirnes PA, Sleight P, Viigimaa Yard, Waeber B, Zannad F, Redon J, Dominiczak A, Narkiewicz K, Nilsson PM, Burnier M, Viigimaa G, Ambrosioni East, Caufield M, Coca A, Olsen MH, Schmieder RE, Tsioufis C, van de Borne P, Zamorano JL, Achenbach S, Baumgartner H, Bax JJ, Bueno H, Dean 5, Deaton C, Erol C, Fagard R, Ferrari R, Hasdai D, Hoes AW, Kirchhof P, Knuuti J, Kolh P, Lancellotti P, Linhart A, Nihoyannopoulos P, Piepoli MF, Ponikowski P, Sirnes PA, Tamargo JL, Tendera M, Torbicki A, Wijns W, Windecker S, Cloudless DL, Coca A, Gillebert TC, Tendera G, Rosei EA, Ambrosioni E, Anker SD, Bauersachs J, Hitij JB, Caulfield M, De Buyzere M, De Geest Southward, Derumeaux GA, Erdine S, Farsang C, Funck-Brentano C, Gerc V, Germano G, Gielen Due south, Haller H, Hoes AW, Jordan J, Kahan T, Komajda M, Lovic D, Mahrholdt H, Olsen MH, Ostergren J, Parati G, Perk J, Polonia J, Popescu BA, Reiner Z, Ryden L, Sirenko Y, Stanton A, Struijker-Boudier H, Tsioufis C, van de Borne P, Vlachopoulos C, Volpe M, Forest DA . 2013 ESH/ESC guidelines for the direction of arterial hypertension: the Task Force for the Direction of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Lodge of Cardiology (ESC). Eur Center J 2013; 34: 2159–2219.
-
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R . Surviving Sepsis Entrada Guidelines Commission including the Pediatric Southward. Surviving sepsis entrada: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 2013; 41: 580–637.
-
Corea 50, Bentivoglio M, Verdecchia P . Echocardiographic left ventricular hypertrophy as related to arterial pressure and plasma norepinephrine concentration in arterial hypertension. Reversal by atenolol treatment. Hypertension 1983; 5: 837–843.
-
Mittal SR . Hateful arterial pressure level may affect LV mass even when dispensary BP is 120/lxxx mm Hg or less. J Assoc Physicians India 2013; 61: 713–715.
-
Atas Northward, Erten Y, Okyay GU, Inal Due south, Topal S, Onec K, Akyel A, Celik B, Tavil Y, Bali Thousand, Arinsoy T . Left ventricular hypertrophy and blood force per unit area control in automated and continuous ambulatory peritoneal dialysis patients. Ther Apher Dial 2014; 18: 297–304.
-
Poyrazoglu HM, Dusunsel R, Yikilmaz A, Narin N, Anarat R, Gunduz Z, Coskun A, Baykan A, Ozturk A . Carotid avenue thickness in children and young adults with terminate stage renal disease. Pediatr Nephrol 2007; 22: 109–116.
-
Maple-Brown 50, Hodge A, Cunningham J, Celermajer DS, O'Dea M . Risk factors for cardiovascular disease practice not fully explain differences in carotid intima-media thickness between Indigenous and European Australians without diabetes. Clin Endocrinol (Oxf) 2009; 71: 189–194.
-
Rogowicz-Frontczak A, Araszkiewicz A, Pilacinski Southward, Zozulinska-Ziolkiewicz D, Wykretowicz A, Wierusz-Wysocka B . Carotid intima-media thickness and arterial stiffness in blazon 1 diabetic patients are dependent on age and mean blood pressure. Exp Clin Endocrinol Diabetes 2011; 119: 281–285.
-
Demirci MS, Gungor O, Kircelli F, Carrero JJ, Tatar E, Demirci C, Kayikcioglu Thou, Asci G, Toz H, Ozkahya Thousand, Ok Due east . Touch on of hateful arterial pressure on progression of arterial stiffness in peritoneal dialysis patients nether strict book control strategy. Clin Nephrol 2012; 77: 105–113.
-
Cypiene A, Dadoniene J, Rugiene R, Ryliskyte L, Kovaite M, Petrulioniene Z, Venalis A, Laucevicius A . The influence of mean blood pressure on arterial stiffening and endothelial dysfunction in women with rheumatoid arthritis and systemic lupus erythematosus. Medicina (Kaunas) 2010; 46: 522–530.
-
Bos WJ, Verrij E, Vincent HH, Westerhof BE, Parati G, van Montfrans GA . How to assess mean blood pressure properly at the brachial avenue level. J Hypertens 2007; 25: 751–755.
Writer data
Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no disharmonize of interest.
Rights and permissions
About this article
Cite this article
Papaioannou, T., Protogerou, A., Vrachatis, D. et al. Mean arterial pressure values calculated using seven different methods and their associations with target organ deterioration in a single-heart study of 1878 individuals. Hypertens Res 39, 640–647 (2016). https://doi.org/10.1038/hour.2016.41
-
Received:
-
Revised:
-
Accepted:
-
Published:
-
Effect Date:
-
DOI : https://doi.org/10.1038/hr.2016.41
Keywords
- aortic stiffness
- carotid artery
- hypertrophy
- left ventricular mass
- pulse-wave velocity
Further reading
Source: https://www.nature.com/articles/hr201641
0 Response to "What Is the Best Way to Estimate the Map?"
Post a Comment